Get Rs 200 off on Oswal Premium Plan for your board preparation!
For Class 9th to 12th. Use Coupon Code BOARD200 at checkout.
Table of Contents
Ans. (a) Because the electrons in magnesium are contained in three shells.
Explanation:
Magnesium comes after sodium in the same period. Atoms of both elements have three electron shells (Na : 2, 8, 1 : Mg : 2, 8, 2). But the nuclear charge of sodium is + 11 and that of magnesium is + 12. Hence, the electron shells are pulled inward more strongly in the Mg atom than in Na atom. Hence, the Mg atom is smaller than Na atom.
Ans. (d) Li
Explanation:
Lithium has the least number of electron shells. This is because of its position in the periodic table, which is higher than sodium and potassium.
Explanation:
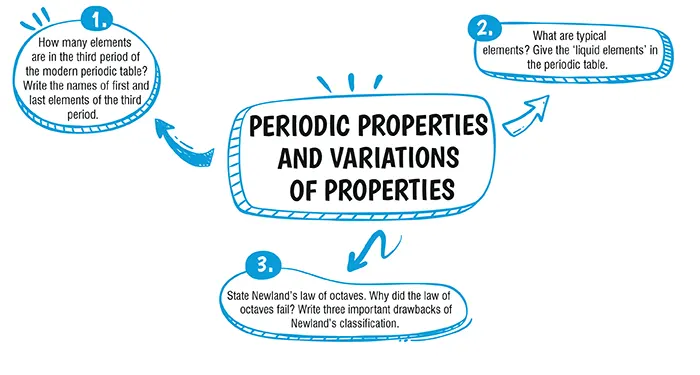
The third period of the periodic table contains eight elements. The first and last elements of the third phase are sodium (Na) and argon (Ar), respectively.
Explanation:
All the elements are found in the third short period of the Periodic Table: Na, Mg, Al, Si, P, S, Cl, and Ar. These components are referred to as typical elements . Liquid elements include bromine, mercury, and francium.
Explanation:
According to Newland's Law of Octaves, an element with a similar property follows every seven elements. similar to how a musical node is repeated inside an octave.The following factors contributed to the law's failure:i The law was only valid up until calcium. Beyond calcium, the other elements could not be included.ii The ninth element, not the eighth, was found to have identical chemical properties with the discovery of rare gases.
Drawbacks:
i Elements could not fit inside the table, according to Newland.
ii Inaccurate estimates of atomic weights were made.
iii Some elements didn't go with the approach.
Download Mind Map of this chapter
Download NowWant to Practice Mock Tests of this chapter
Practice NowDownload Important Questions of this chapter
Download Now| Chapter No. | Chapter Name |
|---|---|
| Chapter 1 | Periodic Properties and Variations of Properties |
| Chapter 2 | Chemical Bonding |
| Chapter 3 | Study of Acids, Bases and Salts |
| Chapter 4 | Analytical Chemistry |
| Chapter 5 | Mole concept and Stoichiometry |
| Chapter 6 | Electrolysis |
| Chapter 7 | Metallurgy |
| Chapter 8 | Study of Compounds : Hydrogen Chloride |
| Chapter 9 | Study of Compounds : Ammonia and Nitric Acid |
| Chapter 10 | Study of Compounds : Sulphuric Acid |
| Chapter 11 | Organic Compounds |
| Chapter Wise Important Questions for ICSE Board Class 10 Chemistry |
|---|
| Periodic Properties and Variations of Properties |
| Chemical Bonding |
| Study of Acids, Bases and Salts |
| Analytical Chemistry |
| Mole concept and Stoichiometry |
| Electrolysis |
| Metallurgy |
| Study of Compounds : Hydrogen Chloride |
| Study of Compounds : Ammonia and Nitric Acid |
| Study of Compounds : Sulphuric Acid |
| Organic Compounds |
CBSE Important Questions Class 10
ICSE Important Questions Class 10
CBSE Important Questions Class 10
ICSE Important Questions Class 10